
Hervé Falet, PhD
Investigator
Hematopoiesis and Immunology

Associate Professor
Department of Cell Biology, Neurobiology and Anatomy
Medical College of Wisconsin
Education and Training
Postdoctoral: Medicine, Brigham and Women's Hospital and Harvard Medical School
PhD: Molecular and Cellular Biology, Paris Descartes University
MS: Molecular Pharmacology, Paris Descartes University
Deficient platelet production, due to genetic causes, secondary to cancer therapy, or from unknown etiology, poses significant risks of mortality, mostly due to bleeding. Our primary research interests are associated with deciphering the cellular mechanisms and proteins regulating normal and abnormal blood platelet production and function.
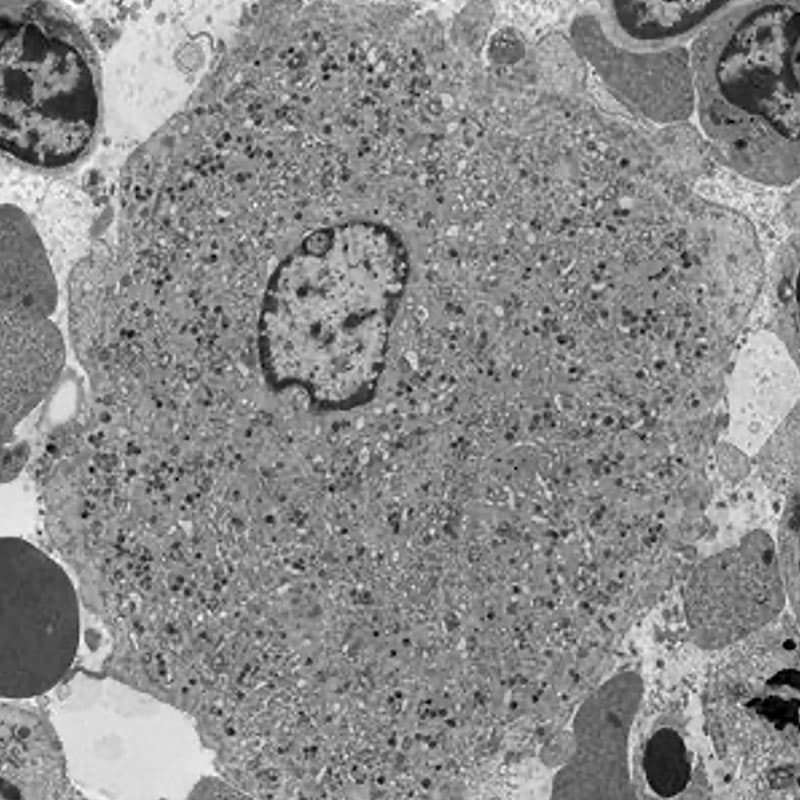
Fig.1. Transmission electron microscopy of a mouse bone marrow megakaryocyte.
Platelet Production
Blood platelets are produced in the bone marrow by megakaryocytes in unique processes that require polyploidisation and extensive membrane rearrangements. These include the formation of the demarcation membrane system, the surface-connected membrane reservoir for future platelets. As megakaryocytes mature, the demarcation membrane system initiates as a single plasma membrane invagination that extends between the lobes of the polyploid nucleus and further expands into its mature form by addition of intracellular membrane materials that are biosynthesized in the endoplasmic reticulum or transported from the Golgi apparatus. The precise molecular mechanisms responsible for these unique membrane rearrangements remain poorly understood. Our current research is focused on the role of receptor-mediated endocytosis in platelet production and function, focusing on the F-BAR protein PACSIN2 and the large GTPase dynamin 2.

Fig.2. α-tubulin (green) and GPIbα (red) immunofluorescence images of a mouse fetal liver–derived megakaryocyte forming proplatelets in vitro.
PACSIN2
PACSIN2 belongs to the F-BAR family of proteins that bind lipid bilayers to generate membrane tubular invaginations in cells. We have identified PACSIN2 as an internal component of the initiating demarcation membrane system in megakaryocytes, where its membrane tabulation activity is regulated by the cytoskeletal and scaffolding protein filamin A (Begonja, Pluthero et al. Blood. 2015;126(1):80-88), a critical regulator of platelet production and function (Falet et al. J Exp Med. 2010;107(9):1967-1979; Begonja et al. Blood. 2011;118(8):2285-2295). Single nucleotide variants in PACSIN2 have recently been associated with altered platelet count and mean platelet volume. We have obtained Pacsin2–/– mice from Dr Markus Plomann (University of Cologne, Germany) to further investigate the role of PACSIN2 in platelet production and function. Pacsin2–/– mice develop mild thrombocytopenia (Biswas, Boyd et al. J Thromb Haemost. 2023 [Epub ahead of print]).
Dynamin 2 (DNM2)
Through its role in membrane fission, dynamin 2 is involved in a wide range of cellular functions, including endocytosis and vesicle transport. DNM2 mutations have been associated with Charcot-Marie-Tooth disease, centronuclear myopathy, and T-cell acute lymphoblastic leukemia in humans and Dnm2 deletion results in early embryonic lethality in mice. We have generated Dnm2fl/fl Pf4-Cre (Dnm2Plt–/–) mice specifically lacking dynamin 2 in the platelet lineage. Dnm2Plt–/– mice develop a severe macrothrombocytopenia (Bender, Giannini et al. Blood. 2015;125(6):1014-1024). Dnm2Plt–/– megakaryocytes accumulate arrested clathrin-coated vesicles that alter their demarcation membrane system, demonstrating that dynamin 2-dependent endocytosis is required for normal megakaryocyte development. Dnm2Plt–/– mice display a profound bleeding diathesis due to impaired platelet hemostatic function (Eaton et al. Haematologica. 2020;105(5):1414-1423).
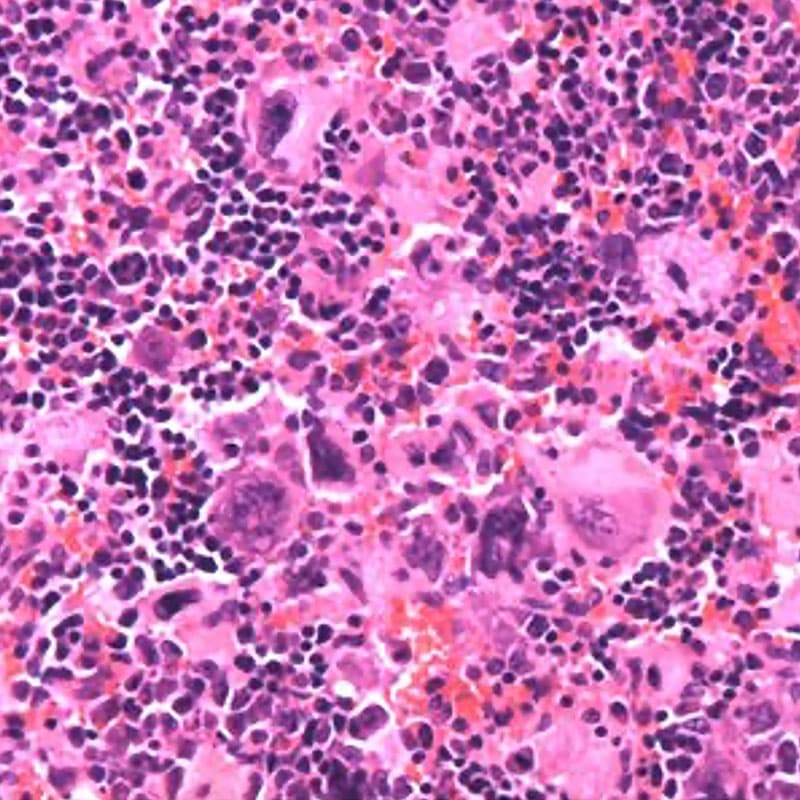
Fig.3. Hematoxylin and eosin staining of Dnm2Plt–/– spleens displaying megakaryocyte hyperplasia.
Myelofibrosis
Dnm2Plt–/– mice also develop hallmarks of myelofibrosis, such as massive megakaryocyte hyperplasia, hematopoietic stem and progenitor cell proliferation, bone marrow fibrosis, extramedullary hematopoiesis, and severe splenomegaly (Bender, Giannini et al. Blood. 2015;125(6):1014-1024). The phenotype is reminiscent of patients with myelofibrosis that is characterized by uncontrolled JAK/STAT signaling in hematopoietic stem cells. At the cellular level, Dnm2Plt–/– platelets display constitutive activation but decreased expression of the tyrosine kinase JAK2 and are unable to endocytose the thrombopoietin receptor Mpl, leading to elevated circulating thrombopoietin levels (Eaton et al. Front Oncol. 2022;12:959806).
In summary, our research addresses the gap of knowledge on receptor-mediated endocytosis in platelet production and function. We anticipate that findings obtained from these studies will yield better understanding to obtain basic science information on how membrane rearrangements contribute to platelet production and function, and to develop treatments for effective re-establishment of platelet production in the setting of thrombocytopenia.
- NIH/NHLBI R01 HL178566 Membrane-cytoskeleton interactions in megakaryocytes and platelets, 08/01/25–04/30/29
Agata Steenackers, PhD
Lab Manager
Mark Zogg
Senior Research Technologist
Emily Boyd, MS
PhD Candidate
Research positions at various levels are available in the Falet lab to study the role of endocytosis in platelet and megakaryocyte biology. To apply, please submit a CV, statement of qualifications, and references to Dr Hervé Falet (HFalet@Versiti.org).
Selected Original Research Articles
Biswas R,* Boyd EK,* Eaton N, Steenackers A, Schulte ML, Reusswig F, Yu H, Drew C, Kahr WHA, Shi Q, Plomann M, Hoffmeister KM, Falet H. PACSIN2 regulates platelet integrin β1 hemostatic function. J Thromb Haemost. 2023 [Epub ahead of print] (*equal contribution) | PMID: 37678551
Eaton N, Boyd EK, Biswas R, Lee-Sundlov MM, Dlugi TA, Ramsey HE, Zheng S, Burns RT, Sola-Visner MC, Hoffmeister KM, Falet H. Endocytosis of the thrombopoietin receptor Mpl regulates megakaryocyte and erythroid maturation in mice. Front Oncol. 2022;12:959806 | PMID: 36110936
Eaton N, Subramaniam S, Schulte ML, Drew C, Jakab D, Haberichter SL, Weiler H, Falet H. Bleeding diathesis in mice lacking JAK2 in platelets. Blood Adv. 2021;5(15):2969-2981 | PMID: 34342643
Unsworth AJ, Bye AP, Sage T, Gaspar RS, Eaton N, Drew C, Stainer A, Kriek N, Volberding PJ, Hutchinson JL, Riley R, Jones S, Mundell SJ, Cui W, Falet H, Gibbins JM. Anti-platelet properties of Pim kinase inhibition is mediated through disruption of thromboxane A2 receptor signalling. Haematologica. 2021;106(7):1968-1978 | PMID: 32467143
Giannini S, Lee-Sundlov MM, Rivadeneyra L, Di Buduo CA, Burns R, Lau JT, Falet H, Balduini A, Hoffmeister KM. β4GalT1 controls β1 integrin function to govern thrombopoiesis and hematopoietic stem cell homeostasis. Nat Commun. 2020;11:356 | PMID: 31953383
Eaton N, Drew C, Wieser J, Munday AD, Falet H. Dynamin 2 is required for GPVI signaling and platelet hemostatic function in mice. Haematologica. 2020;105(5):1414-1423 | PMID: 31296575
Begonja AJ,* Pluthero FG,* Suphamungmee W, Giannini S, Christensen H, Leung R, Lo R, Nakamura F, Lehman W, Plomann M, Hoffmeister KM, Kahr WHA, Hartwig JH, Falet H. FlnA binding to PACSIN2 F-BAR domain regulates membrane tubulation in megakaryocytes and platelets. Blood. 2015;126(1):80-88 (*equal contribution) | PMID: 25838348
Grozovsky R, Begonja AJ, Liu K, Visner G, Hartwig JH, Falet H,* Hoffmeister KM.* The Ashwell-Morell receptor regulates hepatic thrombopoietin production via JAK2-STAT3 signaling. Nat Med. 2015;21(1):47-54 (*equal contribution) | PMID: 25485912
Bender M,* Giannini S,* Grozovsky R, Jönsson T, Christensen H, Pluthero FG, Ko A, Mullally A, Kahr WHA, Hoffmeister KM, Falet H. Dynamin 2-dependent endocytosis is required for normal megakaryocyte development in mice. Blood. 2015;125(6):1014-1024 (*equal contribution) | PMID: 25468568
Begonja AJ, Hoffmeister KM, Hartwig JH, Falet H. FlnA-null megakaryocytes prematurely release large and fragile platelets that circulate poorly. Blood. 2011;118(8):2285-2295 | PMID: 21652675
Falet H, Pollitt AY, Begonja AJ, Weber SE, Duerschmied D, Wagner DD, Watson SP, Hartwig JH. A novel interaction between FlnA and Syk regulates platelet ITAM-mediated receptor signaling and function. J Exp Med. 2010;107(9):1967-1979 | PMID: 20713593
Falet H, Marchetti MP, Hoffmeister KM, Massaad MJ, Geha RS, Hartwig JH. Platelet-associated IgAs and impaired GPVI responses in platelets lacking WIP. Blood. 2009;114(21):4729-4737 | PMID: 19692704
Falet H, Chang G, Brohard-Bohn B, Rendu F, Hartwig JH. αIIbβ3 signals lead cofilin to accelerate platelet actin dynamics. Am J Physiol Cell Physiol. 2005;289(4):C819-C825 | PMID: 15901596
Hoffmeister KM, Felbinger TW, Falet H, Denis CV, Bergmeier W, Mayadas TN, von Andrian UH, Wagner DD, Stossel TP, Hartwig JH. The clearance mechanism of chilled blood platelets. Cell. 2003;112(1):87-97 | PMID: 12526796
Falet H, Hoffmeister KM, Neujahr R, Italiano JE Jr, Stossel TP, Southwick FS, Hartwig JH. Importance of free actin filament barbed ends for Arp2/3 complex function in platelets and fibroblasts. Proc Natl Acad Sci U S A. 2002;99(26):16782-16787 | PMID: 12464680
Falet H, Hoffmeister KM, Neujahr R, Hartwig JH. Normal Arp2/3 complex activation in platelets lacking WASp. Blood. 2002;100(6):2113-2122 | PMID: 12200375
Falet H, Barkalow KL, Pivniouk VI, Barnes MJ, Geha RS, Hartwig JH. Roles of SLP-76, phosphoinositide 3-kinase, and gelsolin in the platelet shape changes initiated by the collagen receptor GPVI/FcRγ-chain complex. Blood. 2000;96(12):3786-3792 | PMID: 11090061
Selected Review Articles
Falet H, Rivadeneyra L, Hoffmeister KM. Clinical impact of glycans in platelet and megakaryocyte biology. Blood. 2022;139(22):3255-3263 | PMID: 35015813
Li R, Hoffmeister KM, Falet H. Glycans and the platelet life cycle. Platelets. 2016;27(6):505-511 | PMID: 27135356
Hoffmeister KM, Falet H. Platelet clearance by the hepatic Ashwell-Morrell receptor: mechanisms and biological significance. Thromb Res. 2016;141(S2):S68-S72 | PMID: 27207430
Grozovsky R, Giannini S, Falet H, Hoffmeister KM. Regulating billions of blood platelets: glycans and beyond. Blood. 2015;126(16):1877-1884 | PMID: 26330242
Falet H. New insights into the versatile roles of platelet FlnA. Platelets. 2013;24(1):1-5 | PMID: 22372530
Selected Book Chapters
Giannini S, Falet H, Hoffmeister KM. Platelet glycobiology and the control of platelet function and lifespan. In: Platelets. Michelson AD, Cattaneo M, Frelinger AL, Newman PJ, Editors. Academic Press, Cambridge; 2019. pp.79-97 | ISBN: 9780128134566
Falet H. Anatomy of the platelet cytoskeleton. In: Platelets in Thrombotic and Non-Thrombotic Disorders: Pathophysiology, Pharmacology and Therapeutics: an Update. Gresele P, Kleiman NS, López JA, Page CP, Editors. Springer, New York; 2017. pp.139-156 | ISBN: 9783319474625
Hervé’s Bibliography on MyNCBI
https://www.ncbi.nlm.nih.gov/myncbi/1hI2on5sGApAz/bibliography/public/