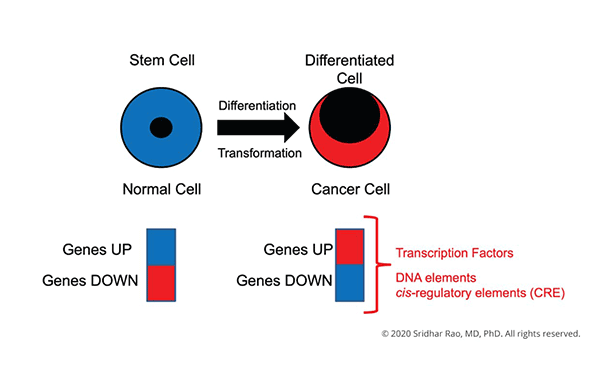
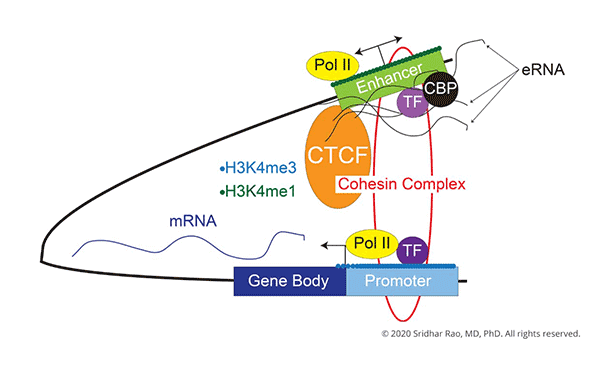
Sridhar
Rao, MD, PhD
Senior Investigator, Program Co-Leader, and Interim Co-Director
Hematopoiesis and Immunology
Senior Investigator, Program Co-Leader, and Interim Co-Director
Associate Professor
Department of Cell Biology, Neurobiology and Anatomy, Medical College of Wisconsin
Education and training
Postdoctoral Research Training
Dana Farber Cancer Institute and Children’s Hospital, Boston, MA
Clinical Training
Dana Farber Cancer Institute and Children’s Hospital, Boston, MA