16S microbiome in IL-10-deficient mice versus wild-type mice
Ray, A., S. Basu, R.Z. Gharaibeh, L.C. Cook, R. Kumar, E.J. Lefkowitz, C.R. Walker, C.D. Morrow, C.L. Franklin, T.L. Geiger, N.H. Salzman, A.F. Fodor and B.N. Dittel. 2015.
Gut microbioal dysbiosis due to Helicobacter drives and increase in marginal zone B cell in the absence of IL-10 signaling in macrophages. J. Immunol. 195:3071-3085.
The metagenomes presented in this article have been submitted to the MG-RAST
Metagenomics Analysis Server (http://metagenomics.anl.gov/linkin.cgi?project=14383)
under MG-RAST identification numbers 4643675.3–4643710.3.
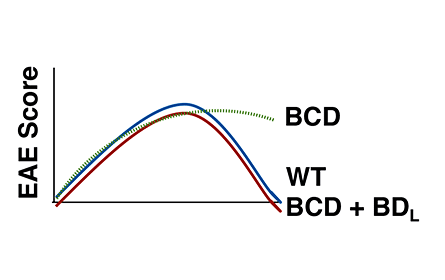
RNAseq in splenic follicular, marginal zone and BDL B cell subsets
Ray, A., Khalil, M., Pulakanti, K.L., Burns, R.T., Gurski, C.J., Basu, S., Wang, D., Rao, S. and Dittel, B.N. 2019.
Mature IgDlow/- B cells maintain tolerance by promoting regulatory T cell homeostasis. Nat. Commun. doi.org/10.1038/s41467-018-08122-9
The RNA-seq data have been deposited in the Gene Expression Omnibus under the accession code GSE111911 and went public December 3, 2018 (https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE111911).