Prithu
Sundd, PhD
Senior Investigator
Thrombosis and Hemostasis
Senior Investigator
Professor of Biomedical Engineering
Professor of Medicine-Division of Hematology and Oncology
Medical College of Wisconsin
Education and Training
Graduate Training
Ohio University, Doctor of Philosophy
Postgraduate Training
La Jolla Institute of Immunology, Postdoctoral Fellow
Prithu Sundd Laboratory at Versiti Blood Research Institute
Sickle cell disease (SCD) is an autosomal recessive genetic disorder that affects more than 100,000 Americans and millions worldwide. Sickle cell anemia (SCA) is the most common form of SCD, which is caused by a substitution of hydrophobic valine (Val) for hydrophilic glutamic acid (Glu) at the sixth position in the beta globin chain. Under hypoxic conditions, the mutated hemoglobin (HbS) undergoes reversible polymerization to form long fibers, which cause red blood cells (RBCs) to sickle. It is believed that sickle RBCs (sRBCs) get trapped in small blood vessels, along with leukocytes and platelets, to cause vaso-occlusion, the predominant pathophysiology responsible for acute pain crises and emergency medical care among SCD patients.
Acute chest syndrome (ACS) is a form of acute lung injury (ALI) and a leading cause of morbidity among SCD patients. SCD patients hospitalized with acute pain crises often develop ACS within the next few days, suggesting a role for vaso-occlusion in lung injury. However, the exact etiological mechanism that triggers ACS is still elusive and the available treatment is primarily supportive.
The Sundd lab aims to elucidate the molecular and biophysical mechanism of leukocyte-platelet-endothelium interaction during inflammation and how these events contribute to vaso-occlusive crisis (VOC) and ACS in SCD. To achieve this, we are using a multi-scale integrative physiologic approach, which involves in vivo multi-photon excitation (MPE) fluorescence microscopy in transgenic and knock-in mice, microfluidic assays with patient blood, total internal reflection fluorescence (TIRF) microscopy, structured illumination microscopy (SIM), laser confocal microscopy, electron microscopy and various biochemical approaches.
This multi-scale approach enables us to address the link between the pathophysiology of ACS affecting the lung (macro-level response) to the aberrant cellular events (micro-level response) driving the vaso-occlusion, and the molecular interactions (nano-level response) that enable those cellular events. Identifying the molecular mechanism of vaso-occlusion in the lung will inspire therapeutics to prevent ACS in SCD patients.
Research Interests
Pulmonary Thrombo-inflammation in Sickle Cell Disease
Sickle cell disease (SCD) affects more than 100,000 Americans and millions more worldwide. Vaso-occlusion, or blockage of blood vessels by blood cell aggregates, is the predominant pathophysiology in SCD. Acute systemic, painful vaso-occlusive episodes, which are the primary reasons for emergency medical care among SCD patients, is often an antecedent to acute chest syndrome (ACS), a type of acute lung injury. ACS is among the leading causes of mortality in SCD, but the current treatment for ACS is primarily supportive, and the etiological mechanism remains largely unknown.
The Sundd Lab uses a multiscale-integrative-physiologic approach involving multi-photon-excitation intravital (in vivo) microscopy of intact lung in live, transgenic, humanized SCD mice and SCD patient blood flowing through a microfluidic platform in vitro.
We have found (Bennewitz et al, JCI-Insight 2017, Bennewitz et al Blood Advances 2020, Vats et al Experimental Hematology 2020) that vaso-occlusive crisis triggered entrapment of P-selectin dependent platelet-neutrophil embolic aggregates in pulmonary arterioles, leading to the arrest of blood flow in the lung of SCD mice. Our recent work (Vats et al, AJRCCM 2020) identifies a role for platelet-inflammasome and IL-1β carrying platelet extracellular vesicles in promoting lung vaso-occlusion in SCD. Our findings suggest that inhibitors of inflammasome or IL-1β dependent innate immune pathway can be beneficial in SCD. Our more recent findings (Vats and Kaminski et al, Blood 2022) show how sterile inflammation in SCD promotes Gasdermin-D-dependent shedding of neutrophil extracellular traps (NETs) in the liver and how these NETs travel intravascularly (embolize) from the liver to the lung, to promote P-selectin-independent lung vascular vaso-occlusion in SCD.
Movie 1
Pulmonary vaso-occlusions (white circles) blocking all 4 arteriolar bottle-necks in a SCD mouse administered 0.1 μg/kg IV LPS. Neutrophil vaso-occlusions (red arrows). Platelet vaso-occlusions (green arrows). White arrow-direction of blood flow. Pulmonary microcirculation (purple). 2/3x original acquisition rate. Movie captured using quantitative Fluorescence Intravital Lung Microscopy (qFILM). Bennewitz and Jimenez et al, Journal of Clinical Investigation Insight. 2017;2(1):e89761.
Movie 2
Neutrophils (red) bound to platelets (blue) are occluding the arteriolar bottleneck in a SCD mouse administered 0.1 μg/kg IV LPS. Erythrocytes (green) are stationary downstream of the vaso-occlusion but erythrocytes upstream of the vaso-occlusion are colliding with the aggregate and then bypassing through the side branch of the arteriole. White arrow-direction of blood flow. Pulmonary microcirculation (purple). 1/3x original acquisition rate. Movie captured using quantitative Fluorescence Intravital Lung Microscopy (qFILM). Bennewitz and Jimenez et al, Journal of Clinical Investigation Insight. 2017;2(1):e89761.
Movie 3
Lung intravital microscopy reveals neutrophil extracellular traps (NETs) in the pulmonary arteriole bottle-neck of an SCD mouse IV administered 10 µmole/kg oxy-Hb. Pulmonary microcirculation (purple), neutrophils (blue), extracellular DNA (green), and citrullinated histones (red). Arrow denotes direction of blood flow. Scale bar 20 µm. 1/3x original acquisition rate. Movie captured using quantitative Fluorescence Intravital Lung Microscopy (qFILM). Vats R and Kaminski TW et al, Blood. 2022 Sep 1;140(9):1020-1037. doi: 10.1182/blood.2021014552.
Movie 4
Lung intravital microscopy reveals circulating NETs (green) entering the lung microcirculation (purple) via the pulmonary arteriole in an SCD mouse IV administered 10 µmole/kg oxy-Hb. Neutrophils (red) and extracellular DNA (green). Arrow denotes direction of blood flow. Scale bar 20 µm. 1/3x original acquisition rate. Movie captured using quantitative Fluorescence Intravital Lung Microscopy (qFILM). Vats R and Kaminski TW et al, Blood. 2022 Sep 1;140(9):1020-1037. doi: 10.1182/blood.2021014552.
Molecular Mechanism of Hemolysis-induced Pulmonary Thrombosis
Patients with hereditary or acquired hemolytic anemias have a high risk of developing in situ pulmonary arteriole thrombosis (iPAT). While pulmonary thrombosis is a major morbidity risk associated with hemolytic disorders, the etiological mechanism underlying hemolysis-induced pulmonary thrombosis remains largely unknown. Recently (Brzoska et al JCI-Insight 2020), we used intravital lung microscopy in mice to assess the pathogenesis of pulmonary thrombosis following deionized water–induced, acute intravascular hemolysis. Acute hemolysis triggered the development of αIIbβ3-dependent, platelet-rich thrombi in precapillary pulmonary arterioles, which led to the transient impairment of pulmonary blood flow.
Consistent with a mechanism involving ADP release from hemolyzing erythrocytes, the inhibition of platelet P2Y12 purinergic receptor signaling attenuated pulmonary thrombosis and rescued blood flow in the pulmonary arterioles of mice following intravascular hemolysis. These findings were the first in vivo studies to suggest that acute intravascular hemolysis promotes ADP-dependent platelet activation, leading to thrombosis in the precapillary pulmonary arterioles. Currently, we are conducting further studies to understand the role of platelet purinergic signaling in the pathogenesis of hemolysis-induced pulmonary thrombosis.
Movie 5
Transient pulmonary thrombosis in WT mouse following 2.5 mg/kg IV ADP. Platelets (green) and pulmonary microcirculation (purple). t = 0 s corresponds to time before and t > 0 s correspond to time following IV ADP administration, respectively. White arrow denotes direction of blood flow within the feeding arteriole. 1.5x original acquisition rate. Movie captured using quantitative Fluorescence Intravital Lung Microscopy (qFILM). Brzoska et al, JCI Insight. 2020 Jul 23;5(14):e139437. doi: 10.1172/jci.insight.139437.
Cigarette Smoke-induced Flu Severity
Cigarette smoking has been associated with the development of flu-induced acute lung injury (ALI). However, the innate immune pathways that leave cigarette smokers at risk of developing flu-triggered ALI remain poorly understood.

Cigarette smoking has been associated with the development of flu-induced acute lung injury (ALI). However, the innate immune pathways that leave cigarette smokers at risk of developing flu-triggered ALI remain poorly understood. We have developed a two-hit model in mice that involves cigarette smoke exposure, followed by intranasal instillation of influenza flu virus. We are using intravital microscopy of lung in mice, biochemical approaches and in vitro studies with patient blood samples to identify the role of neutrophil-platelet aggregates and how innate immune signaling in these cells contributes to the pathogenesis of cigarette smoke-induced severity of flu infection.
In situ formation of large platelet-rich NPAs promotes pulmonary thrombo-inflammation in CS+Flu mice.
Wild type (WT) mice were exposed to cigarette smoke (CS) or room air (RA) for 4 weeks followed by intranasal inoculation with influenza A virus (Flu) and quantitative fluorescence intravital lung microscopy (qFILM) was used to assess thrombo-inflammation in the lung of live mice at 4 days post flu infection. The microcirculation (pseudo-colored purple), neutrophils (red) and platelets (pseudo-colored green) were visualized in vivo by IV administration of FITC dextran, AF546-anti-mouse Ly6G Ab and V450-anti-mouse CD49b Ab, respectively. A neutrophil-bound to platelets (arrowhead) crawls intravascularly towards a large NPA (dashed ellipse) and joins the NPA at 10 min. Scale bar 25 µm. Kaminski TW et al, JCI Insight. 2023 Dec 7:e167299. doi: 10.1172/jci.insight.167299.
Thrombo-inflammatory Mechanism of Joint Damage in Hemophilia
Hemarthrosis (joint bleeding) is a major complication of hemophilia and ultimately leads to debilitating, painful hemophilic arthropathy, primarily affecting elbow, knee and ankle joints. Despite the development and implementation of factor replacement therapies that prevent acute joint bleeding, these events continue to occur, and the current therapy is limited to target joint-replacement surgery.
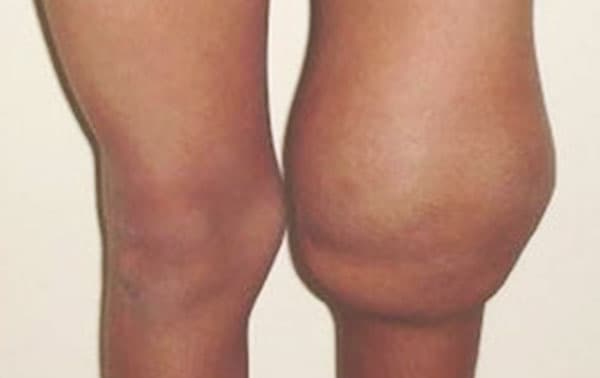
The Sundd Lab is conducting in vitro studies with hemophilia A patient blood samples and in vivo studies in FVIII deficient (hemophilia A) mice to understand the role of inflammasome-dependent, innate immune signaling in promoting hemophilic arthropathy. We hypothesize that bleeding of a synovial joint results in local hemolysis with heme-driven inflammation and ultimate joint destruction, and that therapies targeting inflammasome pathway will alleviate the joint inflammation and prevent long-term joint damage. Currently, experiments are underway to identify the role of eDAMPs (heme and hemoglobin) mediated activation of TLR4 and inflammasome pathway in promoting IL-1b generation, neutrophil-platelet aggregation, neutrophil extracellular traps (NETs) release and progression of joint injury in hemophilia A mice. Based on the preliminary findings, Dr. Sundd recently received the prestigious 2021 Bayer Hemophilia Research Award from the Bayer Hemophilia Award Program.
Techniques and Approaches
Techniques and and approaches used in lab:
- Multi-Photon-Excitation fluorescence intravital (in vivo) microscopy of thrombo-inflammation in live mice.
- Ex vivo microfluidic studies with patient blood.
- Characterization and isolation of extracellular vesicles.
- Knock-in humanized and fluorescent reporter mice of hematological diseases.
- Genomics and transcriptomics.
Lab Team

Tomasz W. Kaminski, PhD
Research Scientist II
Thrombosis and Hemostasis Program
Tomasz W. Kaminski earned his doctoral degree from the Medical University of Bialystok, Poland. During his PhD training, he focused on studying hemostasis disturbances in chronic kidney disease. He joined the Sundd Lab as a postdoctoral associate in 2019. Dr. Kaminski’s research centers on innate immune mechanisms in platelets and neutrophils, as well as the pathophysiology of thrombo-inflammation. He employs state-of-the-art intravital microscopy techniques to capture real-time interactions between neutrophils and platelets during the initial phases of immune system activation. His work exhibits a truly interdisciplinary nature, as he investigates the neutrophil and platelet biology in hemophilic arthropathy, sickle cell disease and influenza. Dr. Kaminski has been recognized with numerous awards from both national and international societies and institutions. Furthermore, his research endeavors receive support from external funding sources.

Manikanta, PhD
Postdoctoral Fellow
Thrombosis and Hemostasis Program
Dr. Manikanta earned his master’s and Ph.D. degree in Biochemistry from University of Mysore. During his Ph.D. time, he worked on the role of platelets in hemostasis and thrombosis. In particular, the role of Apoptosis signal regulating kinase-1 (ASK1) in regulating the platelets functions. He was awarded Ph.D. degree in 2024 and soon after he joined Versiti Blood Research Institute as Postdoctoral fellow. Dr. Manikanta’s current research focus on studying the role of platelets in acute chest syndrome and lung dysfunction in sickle cell disease (SCD). He is also learning the intravital microscopy technique for in vivo imaging of intact murine lung to understands pathophysiology of thrombo-inflammation in humanized SCD mice.

Ayyanar Sivanantham, PhD
Postdoctoral Fellow
Thrombosis and Hemostasis Program
Dr. Sivanantham earned a Master of Technology in Biotechnology and a PhD from Anna University, Chennai, India, where his research focused on therapeutic approaches against acute lung injury and the influence of M1-macrophages on epithelial-mesenchymal transition (EMT).Following his PhD, he completed a three-year postdoctoral training experience at Pulmonary Center, Boston University School of Medicine, where he specialized in the research of lung injury, inflammation, and the role of extracellular vesicles and exosomes. Currently, his research focuses on the functions of extracellular vesicles in neutrophils, particularly under the conditions associated with sickle cell disease during acute chest syndrome.

Nicholas Swendrowski
Senior Research Technician
Thrombosis and Hemostasis Program
Nik attended the University of Wisconsin-La Crosse, where he earned his bachelor's and master's degrees in microbiology. He worked for 10 years in the clinical setting at Wisconsin Diagnostic Laboratory prior to joining Versiti Blood Research Institute.

Marta Wolosowicz, PhD, MBA
Research Scientist I
Thrombosis and Hemostasis Program
Marta Wolosowicz, PhD, MBA, joined Versiti as a Research Scientist I in Dr. Sundd Lab. She earned her doctoral degree from the Medical University of Bialystok in Poland, where her research focused on antioxidant-based interventions in obesity and type 2 diabetes. Her PhD project explored the effects of N-acetylcysteine and α-lipoic acid on fatty acid transport and lipid metabolism in adipose tissue under conditions of metabolic stress, providing new insights into redox-driven regulation of lipid homeostasis. Dr. Wolosowicz also holds a Master of Business Administration in Healthcare Management from the Wroclaw University of Economics and Business. She completed postdoctoral training at the Pittsburgh Institute for Neurodegenerative Diseases, where she studied the mechanisms underlying triosephosphate isomerase deficiency - a rare neuromuscular disorder - and evaluated potential therapeutic strategies. Her PhD project explored the effects of N-acetylcysteine and α-lipoic acid on fatty acid transport and lipid metabolism in adipose tissue under conditions of metabolic stress, providing new insights into redox-driven regulation of lipid homeostasis. Currently, at Versiti, her research focuses on liver senescence and vascular injury in sickle cell disease (SCD), including hemolysis-induced endothelial dysfunction and microvascular occlusion. Her long-term goal is to develop novel therapeutic strategies aimed at mitigating vascular damage in hemolytic disorders.
Recent Sundd Lab Alumni
Margaret Bennewitz, PhD
Former postdoctoral mentee (2013-2017)
Current position: Associate Professor (tenured) of Biomedical Engineering, West Virginia University, Morgantown, WV.
Tomasz Brzoska, PhD
Former postdoctoral mentee (2016-2023)
Current position: Assistant Professor of Medicine-Hem/Onc, University of Pittsburgh School of Medicine, Pittsburgh, PA.
Ravi Vats, PhD
Former bioengineering PhD student (2017-2022)
Current position: Research Operations Manager, DISC Medicine Inc., Watertown, MA
Rikesh K. Dubey, PhD
Former postdoctoral mentee (2021-2025)
Current position: Assistant Professor, Division of Medical Research, Faculty of Medical & Health Sciences, SRM- Institute of Science and Technology, TN, India.
Egemen Tutuncuoglu, MSc Former senior research technician (2014-2022)
Current position: Gladstone Institute, San Francisco, CA
Maritza A. Montanez, PhD Former bioengineering PhD student (2013-2018)
Current position: University of Pittsburgh Medical Center Artificial Heart Program
Omika Katoch, PhD
Former postdoctoral mentee (2021-2025)
Adekunle E. Alagbe, MD, PhD Former postdoctoral mentee (2022-2023)
Current position: University of Pittsburgh School of Medicine.
Grant Support
NHLBI 2R01 HL128297 ” Pulmonary arteriole occlusion by platelet-neutrophil micro-emboli in Acute Chest Syndrome”. Role: Contact PI (2015 – 2025)
NHLBI 1R01 HL166345 “CD39-carrying extracellular vesicles regulate pulmonary thrombosis in Sickle Cell Disease”. Role: Contact PI (2023-2027)
American Heart Association 23TPA1074022 “Thrombo-inflammation in cigarette smoke-induced flu severity”. Role: Contact PI (2023-2026)
CSL Behring Inc Research Acceleration Initiative. Role: Contact PI (2021-2026)
Publications
Selected Publications
- Shaked I, Foo C, Mächler P, Liu R, Cui Y, Ji X, Broggini T, Kaminski T, Suryakant Jadhav S, Sundd P, Firer M, Devor A, Friedman B, Kleinfeld D. A lone spike in blood glucose can enhance the thrombo-inflammatory response in cortical venules. J Cereb Blood Flow Metab. 2023 Sep 22:271678X231203023. doi: 10.1177/0271678X231203023. PubMed PMID: 37737093.
- Dubey RK, Vats R, Brzoska T, Kaminski TW, Katoch O, Tejero J, Njikang G, Paderi J, Sundd P. Subcutaneous injection of IHP-102 prevents lung vaso-occlusion in sickle cell disease mice. Haematologica. 2023 Oct 19. doi: 10.3324/haematol.2023.283716. Online ahead of print. PubMed PMID: 37855052.
- Kaminski TW, Katoch O, Li Z, Hanway CB, Dubey RK, Alagbe A, Brzoska T, Zhang H, Sundd P, Kato GJ, Novelli EM, Pradhan-Sundd T. Impaired hemoglobin clearance by sinusoidal endothelium promotes vaso-occlusion and liver injury in sickle cell disease. Haematologica. 2023 Nov 9. doi: 10.3324/haematol.2023.283792. Online ahead of print. PubMed PMID: 37941440.
- Kaminski TW, Brzoska T, Li X, Vats R, Katoch O, Dubey RK, Bagale K, Watkins SC, McVerry BJ, Pradhan-Sundd T, Zhang L, Robinson KM, Nyunoya T, Sundd P. Lung microvascular occlusion by platelet-rich neutrophil-platelet aggregates promotes cigarette smoke induced severe flu. JCI Insight. 2024 Jan 23;9(2):e167299. doi: 10.1172/jci.insight.167299. PubMed PMID: 38060312.
- Kaminski TW, Sivanantham A, Mozhenkova A, Smith A, Ungalara R, Dubey R, Shrestha B, Hanway C, Katoch O, Tejero J, Sundd P, Novelli EM, Kato GJ, Pradhan-Sundd T. Hemoglobin scavenger receptor CD163 as a potential biomarker of hemolysis induced hepatobiliary injury in sickle cell disease. Am J Physiol Cell Physiol. 2024 Apr 29. doi: 10.1152/ajpcell.00386.2023. PubMed PMID: 38682236.
- Das S, Kaminski TW, Schlegel BT, Bain W, Hu S, Patel A, Kale SL, Chen K, Lee JS, Mallampalli RK, Kagan VE, Rajasundaram D, McVerry BJ, Sundd P, Kitsios GD, Ray A, Ray P. Neutrophils and galectin-3 defend mice from lethal bacterial infection and humans from acute respiratory failure. Nat Commun. 2024 Jun 3;15(1):4724. doi: 10.1038/s41467-024-48796-y. PubMed PMID: 38830855.
- Pai A, Sundd P, Tees DF. In situ Microrheological Determination of Neutrophil Stiffening Following Adhesion in a Model Capillary. Ann Biomed Eng. 2008;36(4):596-603. PubMed PMID: 18214680.
- Sundd P, Zou X, Goetz DJ, Tees DF. Leukocyte adhesion in capillary-sized, P-selectin-coated micropipettes. Microcirculation. 2008;15(2):109-122. PubMed PMID: 17952800.
- Sundd P, Gutierrez E, Pospieszalska MK, Zhang H, Groisman A, Ley K. Quantitative dynamic footprinting microscopy reveals mechanisms of neutrophil rolling. Nature Methods. 2010;7(10):821-824. PubMed PMID: 20871617; PubMed Central PMCID: PMC2967732. Images from this study were on the cover page of the October, 2010 issue of Nature Methods.
- Gutierrez E, Tkachenko E, Besser A, Sundd P, Ley K, Danuser G, Ginsberg MH, Groisman A. High refractive index silicone gels for simultaneous total internal reflection fluorescence and traction force microscopy of adherent cells. PLoS One. 2011;6(9):e23807. PubMed PMID: 21961031; PubMed Central PMCID: PMC3178539.
- Sundd P, Gutierrez E, Petrich BG, Ginsberg MH, Groisman A, Ley K. Live cell imaging of paxillin in rolling neutrophils by dual-color quantitative dynamic footprinting. Microcirculation. 2011;18(5):361-372. PubMed PMID: 21418380; PubMed Central PMCID: PMC3123727. Images from this study were on the cover page of the July, 2011 issue of Microcirculation.
- Sundd P, Gutierrez E, Koltsova EK, Kuwano Y, Fukuda S, Pospieszalska MK, Groisman A, Ley K. 'Slings' enable neutrophil rolling at high shear. Nature. 2012;488(7411):399-403. PubMed PMID: 22763437; PubMed Central PMCID: PMC3433404.
- Stadtmann A, Germena G, Block H, Boras M, Rossaint J, Sundd P, Lefort C, Fisher CI, Buscher K, Gelschefarth B, Urzainqui A, Gerke V, Ley K, Zarbock A. The PSGL-1-L-selectin signaling complex regulates neutrophil adhesion under flow. Journal of Experimental Medicine. 2013;210(11):2171-2180. PubMed PMID: 24127491; PubMed Central PMCID: PMC3804951.
- Koltsova EK, Sundd P, Zarpellon A, Ouyang H, Mikulski Z, Zampolli A, Ruggeri ZM, Ley K. Genetic deletion of platelet glycoprotein Ibα but not its extracellular domain protects from atherosclerosis. Thromb Haemost. 2014;112(6):1252-1263. PubMed PMID: 25104056; PubMed Central PMCID: PMC4429870.
- Bennewitz MF, Watkins SC, Sundd P. Quantitative intravital two-photon excitation microscopy reveals absence of pulmonary vaso-occlusion in unchallenged Sickle Cell Disease mice. Intravital. 2014;3(2):e29748. PubMed PMID: 25995970; PubMed Central PMCID: PMC4435611.
- Jimenez MA, Tutuncuoglu E, Barge S, Novelli EM, Sundd P. Quantitative microfluidic fluorescence microscopy to study vaso-occlusion in sickle cell disease. Haematologica. 2015;100(10):e390-393. PubMed PMID: 25975836; PubMed Central PMCID: PMC4591772.
- Bennewitz MF†, Jimenez MA†, Vats R, Tutuncuoglu E, Jonassaint J, Kato GJ, Gladwin MT, Sundd P. Lung vaso-occlusion in sickle cell disease mediated by arteriolar neutrophil-platelet microemboli. Journal of Clinical Investigation Insight. 2017;2(1):e89761. PubMed PMID: 28097236; PubMed Central PMCID: PMC5214368. †Co-first authors.
- Jimenez MA, Novelli E, Shaw GD, Sundd P. Glycoprotein Ibα inhibitor (CCP-224) prevents neutrophil-platelet aggregation in Sickle Cell Disease. Blood Advances. 2017;1(20):1712-1716. PubMed PMID: 28966995; PubMed Central PMCID: PMC5617353. Images from this study were on the cover page of the September 12 issue of Blood Advances.
- Pradhan-Sundd T, Zhou L, Vats R, Jiang A, Molina L, Singh S, Poddar M, Russell JM, Stolz DB, Oertel M, Apte U, Watkins S, Ranganathan S, Nejak-Bowen KN, Sundd P, Pal Monga S. Dual catenin loss in murine liver causes tight junctional deregulation and progressive intrahepatic cholestasis. Hepatology. 2018 Jun;67(6):2320-2337. doi: 10.1002/hep.29585. PubMed PMID: 29023813; PubMed Central PMCID: PMC5893443.
- Lendermon EA, Dodd-O JM, Coon TA, Wang X, Ensor CR, Cardenes N, Koodray CL, Heusey HL, Bennewitz MF, Sundd P, Bullock GC, Popescu I, Guo L, O'Donnell CP, Rojas M, McDyer JF. Azithromycin Fails to Prevent Accelerated Airway Obliteration in T-bet-/- Mouse Lung Allograft Recipients. Transplant Proc. 2018 Jun;50(5):1566-1574. PubMed PMID: 29880387.
- Pradhan-Sundd T, Vats R, Russell JM, Singh S, Michael AA, Molina L, Kakar S, Cornuet P, Poddar M, Watkins SC, Nejak-Bowen KN, Monga SP and Sundd P. Dysregulated bile transporters and impaired tight junctions during chronic liver injury in mice. Gastroenterology. 2018 Oct;155(4):1218-1232.e24. PubMed PMID: 29964040; PubMed Central PMCID: PMC6174089.
- Li ZG, Scott MJ, Brzóska T, Sundd P, Li YH, Billiar TR, Wilson MA, Wang P, Fan J. Lung epithelial cell-derived IL-25 negatively regulates LPS-induced exosome release from macrophages. Mil Med Res. 2018 Jul 30;5(1):24. PubMed PMID: 30056803; PubMed Central PMCID: PMC6065058.
- Fatkhullina AR, Peshkova IO, Dzutsev A, Aghayev T, McCulloch JA, Thovarai V, Badger JH, Vats R, Sundd P, Tang HY, Kossenkov AV, Hazen SL, Trinchieri G, Grivennikov SI, Koltsova EK. An Interleukin-23-Interleukin-22 Axis Regulates Intestinal Microbial Homeostasis to Protect from Diet-Induced Atherosclerosis. Immunity. 2018 Nov 20;49(5):943-957.e9. PubMed PMID: 30389414; PubMed Central PMCID: PMC6257980.
- Voic H, Li X, Jang JH, Zou C, Sundd P, Alder J, Rojas M, Chandra D, Randell S, Mallampalli RK, Tesfaigzi Y, Ryba T, Nyunoya T. RNA sequencing identifies common pathways between cigarette smoke exposure and replicative senescence in human airway epithelia. BMC Genomics. 2019 Jan 9;20(1):22. PubMed PMID: 30626320; PubMed Central PMCID: PMC6325884.
- Vats R†, Brzoska T†, Bennewitz MF†, Jimenez MA†, Pradhan-Sundd T, Tutuncuoglu E, Jonassaint J, Gutierrez E, Watkins SC, Shiva S, Scott M, Morelli AE, Neal MD, Kato GJ, Gladwin MT, Sundd P. Platelet extracellular vesicles drive inflammasome-IL1β-dependent lung injury in Sickle Cell Disease. Am J Respir Crit Care Med. 2020 Jan 1;201(1):33-46. PubMed PMID: 31498653; PubMed Central PMCID: PMC6938158. †Co-first authors.
- Dyer MR, Alexander W, Hassoune A, Chen Q, Alvikas J, Liu Y, Haldeman S, Plautz W, Loughran P, Li H, Boone B, Sadovsky Y, Sundd P, Zuckerbraun BS, Neal MD. Platelet-derived extracellular vesicles released after trauma promote hemostasis but contribute to DVT in mice. 2019. J Thromb Haemost. 2019 Oct;17(10):1733-1745. PubMed PMID: 31294514; PubMed Central PMCID: PMC6773503.
- Pradhan‐Sundd T, Kosar K, Saggi H, Zhang R, Vats R, Cornuet P, Green S, Singh S, Zeng G, Sundd P, Nejak-Bowen K. Wnt/β-catenin signaling plays a protective role in the mdr2 ko murine model of cholestatic liver disease. Hepatology. 2019 Sep 6. doi: 10.1002/hep.30927. PubMed PMID: 31489648.
- Bennewitz MF, Tutuncuoglu E, Gudapati S, Brzoska T, Watkins SC, Monga SP, Pradhan-Sundd T, Sundd P. P-selectin deficient mice to study pathophysiology of Sickle Cell Disease. Blood Adv. 2020 Jan 28;4(2):266-273. PubMed PMID: 31968076; PubMed Central PMCID: PMC6988409. Images from this study were on the cover page of the January 28 issue of Blood Advances.
- Mallampalli RK, Li X, Jang JH, Kaminski T, Hoji A, Coon T, Chandra D, Welty S, Teng Y, Ihle J, Sembrat J, Rojas M, Lafyatis R, Zou C, Sciurba F, Sundd P, Lan L, Nyunoya T. Cigarette Smoke Exposure Enhances Transforming Acidic Coiled-Coil-Containing Protein 2 (TACC2) Turnover and therby Promotes Emphysema. JCI Insight. 2020 Jan 30;5(2). pii: 125895. PubMed PMID: 31996486. PubMed Central PMCID: PMC7098723.
- Sparkenbaugh EM, Chen C, Brzoska T, Nguyen J, Wang S, Vercellotti GM, Key NS, Sundd P, Belcher JD, Pawlinski R. Thrombin-mediated activation of PAR-1 contributes to microvascular stasis in mouse models of sickle cell disease. Blood. 2020 Jan 23. pii: blood.2019003543. doi: 10.1182/blood.2019003543. PubMed PMID: 31977004.
- Vats R, Liu S, Zhu J, Mukhi D, Tutuncuoglu E, Cardenes N, Singh S, Brzoska T, Kosar K, Bamne M, Jonassaint J, Adebayo Michael A, Watkins SC, Hillery C, Ma X, Nejak-Bowen K, Rojas M, Gladwin MT, Kato GJ, Ramakrishnan S, Sundd P, Pal Monga S, Pradhan-Sundd T. Impaired bile acid secretion promotes hepatobiliary injury in Sickle Cell Disease. Hepatology 2020 Mar 19. doi: 10.1002/hep.31239. PubMed PMID: 32190913.
- Vats R, Tutuncuoglu E, Pradhan-Sundd T, Tejero J, Shaw GD, Sundd P. Tandem P-selectin glycoprotein ligand immunoglobulin (TSGL-Ig) prevents lung vaso-occlusion in Sickle Cell Disease mice. Experimental Hematology. 2020 Apr;84:1-6.e1. doi: 10.1016/j.exphem.2020.03.002. PubMed PMID: 32243995. PubMed Central PMCID: PMC7237323.
- Brzoska T, Vats R, Bennewitz MF, Tutuncuoglu E, Watkins SC, Ragni MV, Neal MD, Gladwin MT, Sundd P. Intravascular hemolysis triggers ADP-mediated generation of platelet-rich thrombi in pre-capillary pulmonary arterioles. Journal of Clinical Investigation-Insight. 2020 Jul 23;5(14). doi: 10.1172/jci.insight.139437. PubMed PMID: 32544100; PubMed Central PMCID: PMC7453902.
- Zhao J, Florentin J, Tai YY, Torrino S, Ohayon L, Brzoska T, Tang Y, Yang J, Negi V, Woodcock CC, Risbano MG, Nouraie SM, Sundd P, Bertero T, Dutta P, Chan SY. Long Range Endocrine Delivery of Circulating miR-210 to Endothelium Promotes Pulmonary Hypertension. Circ Res. 2020 Aug 14;127(5):677-692. doi: 10.1161/CIRCRESAHA.119.316398. Epub 2020 Jun 4. PubMed PMID: 32493166; PubMed Central PMCID: PMC7429300.
- Perkins LA, Nyiranshuti L, Little-Ihrig L, Latoche JD, Day KE, Zhu Q, Tavakoli S, Sundd P, Novelli EM, Anderson CJ. Integrin VLA-4 as a PET imaging biomarker of hyper-adhesion in transgenic sickle mice. Blood Adv. 2020 Sep 8;4(17):4102-4112. doi: 10.1182/bloodadvances.2020002642. PubMed PMID: 32882004; PubMed Central PMCID: PMC7479956.
- Crompton D, Vats R, Pradhan-Sundd T, Sundd P, Kameneva MV. Drag-reducing polymers improve hepatic vaso-occlusion in SCD mice. Blood Adv. 2020 Sep 22;4(18):4333-4336. doi: 10.1182/bloodadvances.2020002779. PubMed PMID: 32915976; PubMed Central PMCID: PMC7509886.
- Vats R, Kaminski TW, Ju EM, Brzoska T, Tutuncuoglu E, Tejero J, Novelli EM, Sundd P*, Pradhan-Sundd T. P-selectin deficiency promotes liver senescence in sickle cell disease mice. Blood. 2021 May 13;137(19):2676-2680. doi:10.1182/blood.2020009779. PubMed PMID:33619560. *Co-corresponding author.
- Li X, Noell G, Tabib T, Gregory AD, Trejo Bittar HE, Vats R, Kaminski TW, Sembrat J, Snyder ME, Chandra D, Chen K, Zou C, Zhang Y, Sundd P, McDyer JF, Sciurba F, Rojas M, Lafyatis R, Shapiro SD, Faner R, Nyunoya T. Single cell RNA sequencing identifies IGFBP5 and QKI as ciliated epithelial cell genes associated with severe COPD. Respir Res. 2021 Apr 6;22(1):100. doi: 10.1186/s12931-021-01675-2. PubMed PMID: 33823868. PubMed Central PMCID: PMC8022543.
- Crompton D, Gudla S, Waters JH, Sundd P, Kameneva MV. Hemorheological Approach to Improve Perfusion of Red Blood Cells with Reduced Deformability Using Drag-Reducing Polymer (In Vitro Study). ASAIO J. 2021 Aug 16. doi: 10.1097/MAT.0000000000001559. Online ahead of print. PubMed PMID: 34406139.
- Molina LM, Zhu J, Li Q, Pradhan-Sundd T, Krutsenko Y, Sayed K, Jenkins N, Vats R, Bhushan B, Ko S, Hu S, Poddar M, Singh S, Tao J, Sundd P, Singhi A, Watkins S, Ma X, Benos PV, Feranchak A, Michalopoulos G, Nejak-Bowen K, Watson A, Bell A, Monga SP. Compensatory hepatic adaptation accompanies permanent absence of intrahepatic biliary network due to YAP1 loss in liver progenitors. Cell Rep. 2021 Jul 6;36(1):109310. doi: 10.1016/j.celrep.2021.109310. PubMed PMID: 34233187.
- Giordano L, Gregory AD, Pérez Verdaguer M, Ware SA, Harvey H, DeVallance E, Brzoska T, Sundd P, Zhang Y, Sciurba FC, Shapiro SD, Kaufman BA. Extracellular Release of Mitochondrial DNA: Triggered by Cigarette Smoke and Detected in COPD. Cells 2022. Jan 22;11(3):369. doi: 10.3390/cells11030369. PubMed PMID: 35159179. PMCID: PMC8834490.
- Kaminski TW, Ju EM, Gudapati S, Vats R, Arshad S, Dubey RK, Katoch O, Tutuncuoglu E, Frank J, Brzoska T, Stolz DB, Watkins SC, Chan SY, Ragni MV, Novelli EM, Sundd P, Pradhan-Sundd T. Defenestrated endothelium delays liver-directed gene transfer in hemophilia A mice. Blood Advances. 2022 Jun 28;6(12):3729-3734. doi: 10.1182/bloodadvances.2021006388. PubMed PMID: 35427414.
- Carreño M, Pires MF, Woodcock SR, Brzoska T, Ghosh S, Salvatore SR, Chang F, Khoo NKH, Dunn M, Connors N, Yuan S, Straub AC, Wendell SG, Kato GJ, Freeman BA, Ofori-Acquah SF, Sundd P, Schopfer FJ, Vitturi DA. Immunomodulatory actions of a kynurenine-derived endogenous electrophile. Science Advances. 2022 Jul;8(26):eabm9138. doi:10.1126/sciadv.abm9138. PubMed PMID: 35767602. PMCID: PMC9242454.
- Vats R†, Kaminski TW†, Brzoska T, Leech JA, Tutuncuoglu E, Katoch O, Jonassaint JC, Tejero J, Novelli EM, Pradhan-Sundd T, Gladwin MT, Sundd P. Liver to lung microembolic NETs promote Gasdermin-D-dependent inflammatory lung injury in Sickle Cell Disease. Blood. 2022 Sep 1;140(9):1020-1037. doi: 10.1182/blood.2021014552. PubMed PMID: 35737916. This study was the cover article in the Sep 1, 2022 issue of Blood.
Review Articles
- Gurkan UA, Wood DK, Carranza D, Herbertson LH, Diamond SL, Du E, Guha S, Di Paola J, Hines PC, Papautsky I, Shevkoplyas SS, Sniadecki NJ, Pamula VK,Sundd P, Rizwan A, Qasba P, Lam WA. Next generation microfluidics: fulfilling the promise of lab-on-a-chip technologies. Lab Chip. 2024 Mar 26;24(7):1867-1874. doi: 10.1039/d3lc00796k. PubMed PMID: 38487919.
- Sundd P*, Pospieszalska MK, Cheung LS, Konstantopoulos K, Ley K. Biomechanics of leukocyte rolling. Biorheology. 2011;48(1):1-35. PubMed PMID: 21515934; PubMed Central PMCID: PMC3103268. A figure from this manuscript was on the cover page of Biorheology. *Corresponding author.
- Sundd P, Pospieszalska MK, Ley K. Neutrophil rolling at high shear: flattening, catch bond behavior, tethers and slings. Molecular Immunology. 2013;55(1):59-69. PubMed PMID: 23141302; PubMed Central PMCID: PMC3601566.
- Sundd P, Ley K. Quantitative dynamic footprinting microscopy. Immunology and cell biology. 2013;91(4):311-320. PubMed PMID: 23478358.
- Sundd P, Gladwin MT and Novelli EM. Pathophysiology of sickle cell disease. Annu Rev Pathol. 2019 Jan 24;14:263-292. PubMed PMID: 30332562; PubMed Central PMCID: PMC7053558.
- Fan Z, Mikulski Z, McArdle S, Sundd P, Ley K. Super-STORM: Molecular Modeling to Achieve Single-molecule Localization with STORM Microscopy. STAR Protoc. 2020 Jun 3;1(1):100012. doi: 10.1016/j.xpro.2019.100012. eCollection 2020 Jun 19. PubMed PMID: 33111071.
- Brzoska T, Kaminski TW, Bennewitz MF, Sundd P. Live Imaging of the Lung. Curr Protoc Cytom. 2020 Dec;95(1):e80. doi: 10.1002/cpcy.80. PubMed PMID: 33226733. PubMed Central PMCID: PMC8021006.
- Li G, Chen T, Dahlman J, Eniola-Adefeso L, Ghiran IC, Kurre P, Lam WA, Lang JK, Marbán E, Martín P, Momma S, Moos M, Nelson DJ, Raffai RL, Ren X, Sluijter JPG, Stott SL, Vunjak-Novakovic G, Walker ND, Wang Z, Witwer KW, Yang PC, Lundberg MS, Ochocinska MJ, Wong R, Zhou G, Chan SY, Das S, Sundd P. Current challenges and future directions for engineering extracellular vesicles for heart, lung, blood and sleep diseases. J Extracell Vesicles. 2023 Feb;12(2):e12305. doi: 10.1002/jev2.12305.
- Chiang KC, Gupta A, Sundd P, Krishnamurti L. Thrombo-Inflammation in COVID-19 and Sickle Cell Disease: Two Faces of the Same Coin. Biomedicines 2023 Jan 25;11(2):338. doi: 10.3390/biomedicines11020338. PMID: 36830874 PMCID: PMC9953430.
Editorials
Sundd P and Kuebler W. Smooth muscle cells: A novel site of P-selectin expression with pathophysiological and therapeutic relevance in pulmonary hypertension. Am J Respir Crit Care Med. 2019 Jun 1;199(11):1307-1309. PubMed PMID: 30592637. PMCID: PMC6543715.
Book Chapters
- Tees DFJ, Sundd P, Goetz DJ. A flow chamber for capillary networks: Leukocyte adhesion in capillary-sized, ligand-coated micropipettes. In: Principles of cellular engineering: Understanding the biomolecular interface, edited by M. R. King. New York: Academic Press, 2006.
- Ley K, Mestas J, Pospieszalska MK, Sundd P, Groisman A, and Zarbock A. Intravital microscopic investigation of leukocyte interactions with the blood vessel wall. In: Methods in Enzymology: Elsevier Academic Press, 445, 2008, p. 255-79. PubMed PMID: 19022063.
- Sundd, P. & Bennewitz, M.F. Leukocyte kinetics and migration in the lungs. in Hematologic Abnormalities and Acute Lung Syndromes (eds. Lee, J.S. & Donahoe, M.P.) 19-45 (Springer International Publishing, Switzerland, 2017). 10.1007/978-3-319-41912-1.
- Brzóska T, Kato GJ and Sundd P. The Role of Platelets in Sickle Cell Disease. In: A. D. Michelson, ed. Platelets. Fourth ed.: Elsevier Inc.; 2019: 563-580.
- Sundd P and Ofori-Acquah SF. Sterile inflammation in sickle cell disease. In: Gladwin MT, Kato GJ, Novelli EM, eds. Sickle Cell Disease. New York, NY: McGraw Hill; 2021: 93-108.